Recently, the Jiangsu Key Laboratory of Immunity and Metabolism at our university has officially published a research paper entitled "Multi-omics reveals cholesterol-driven macrophage metabolic reprogramming and inflammation in chronic obstructive pulmonary disease" in Genome Medicine (a top journal in CAS Zone 1, IF = 11.2). Professor Guo Feng from the School of Basic Medicine, Xuzhou Medical University, serves as the corresponding author. This research was supported by grants from the National Natural Science Foundation of China, the Natural Science Foundation of Jiangsu Province, and the Special Program of Guangzhou National Laboratory, among other projects.
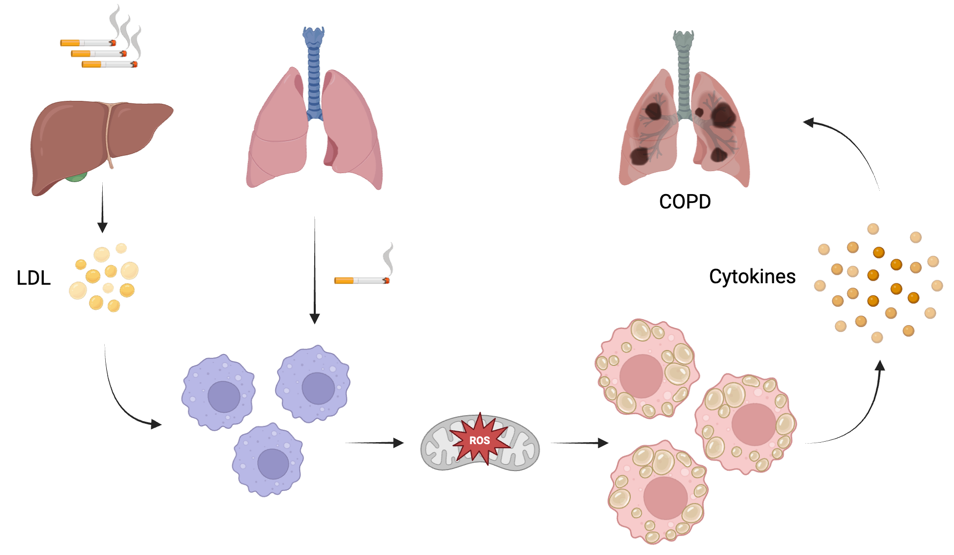
Chronic obstructive pulmonary disease (COPD) is a progressive inflammatory disorder characterized by irreversible airflow limitation, airway remodeling, and alveolar structural destruction. Recent studies have demonstrated that disrupted tissue homeostasis, driven by systemic metabolic abnormalities and chronic inflammation, plays a critical role in the pathogenesis and progression of COPD, among which dyslipidemia is particularly prominent.
In this study, metabolomic analysis of plasma from COPD patients revealed that plasma cholesterol levels were significantly negatively correlated with the predicted percentage of forced expiratory volume in one second (FEV1%) and the FEV1/FVC ratio, and gradually increased with disease severity, suggesting that imbalanced cholesterol metabolism is closely associated with declined lung function. Further results showed that cigarette smoke and cholesterol acted synergistically to markedly impair mitochondrial function in alveolar macrophages, induce excessive reactive oxygen species (ROS) production, and upregulate the expression of Peptidylprolyl isomerase A (PPIA). These events subsequently activated the NF-κB signaling pathway, promoted the release of IL-1β, and thereby amplified pulmonary inflammatory responses.
By integrating metabolomic analysis, a mouse model fed a high-cholesterol diet, and mechanistic studies of macrophage function, this study confirmed that dysregulated cholesterol metabolism represents a key metabolic feature of COPD and actively contributes to disease progression. It also uncovered the pivotal role of the cholesterol–ROS–PPIA–NF-κB inflammatory axis in COPD-related inflammation and tissue damage, indicating that targeting cholesterol metabolism or intervening in mitochondrial homeostasis may serve as potential therapeutic strategies to slow the progression of COPD.
Article link: https://link.springer.com/article/10.1186/s13073-025-01591-w

(First Review: Li Li Second Review: Wang Wenshi Third Review: Han Hongliu)